Energy and Place Project:
Essential Questions:
- How does energy production and consumption impact place?
- How do your sense of place, environmental ethic and understanding of our energy needs influence your perception and decisions relating to energy production and consumption?
Reflection:
From completing the capstone energy lab I learned some interesting things about science and its processes in the real world. The first thing that I learned about science is that the processes that are used to come to amazing understandings are sometimes very tedious or even sometimes seem very simple. For instance all that Liam and I had to do to watch the half life curve of a radionuclide was hook up a computer graphing software to a geiger counter measuring the counts of decay for a barium-137. This seemed easy but to understand the concept of tiny beyond belief particles shooting from a substance until that substance becomes stable maybe in minutes or maybe in millions of years is amazing. This is the thing that I have come to appreciate about science, at a first glance the mol may seem like a tedious conversion process with a lot of zeros, but when you have a chance to stop and think about it you real in the wonder of how enormously eternal the pieces that make up the universe are. All in all processes can be interesting or they can be mind straining but the results are so important to humanity.
Through my scientific paper I learned that nuclear energy is very powerful. I learned that if we wish to use it at a greater scale in the future there are a lot of decisions that lay on our hands. One of these disiions for instance is that we need to look into the effects of elements with long half lives that will contaminate what is around them for a long time versus elements with short half lives that are very dangerous for a short amount of time. Through my infographic I learned that learned the amount of Ionizing radiation we have been exposed to has gone up by a considerable amount from medical procedures involving radiation. Here is another example of dilemma that nuclear power provides humanity with. Something that causes cancer can also help heal us from it.
Through my infographic I was trying to show people that we are exposed to a considerably higher amount of radiation then we were in 1987. I wanted to convey this so that people can understand not only what Ionizing radiation is, and what its effects are but also the reality of how we use it and what actions we are taking with it that both benefit and harm humanity. I wanted to provide an accurate representation of the Ionizing radiation sources we are exposed to. I wanted to show the benefits and the drawbacks of nuclear medicine and radiation therapy because those are the greatest growing categories of our exposure. I wanted to do this all so that people could have more understanding about our world and our interaction with it through our technologies.
From completing the capstone energy lab I learned some interesting things about science and its processes in the real world. The first thing that I learned about science is that the processes that are used to come to amazing understandings are sometimes very tedious or even sometimes seem very simple. For instance all that Liam and I had to do to watch the half life curve of a radionuclide was hook up a computer graphing software to a geiger counter measuring the counts of decay for a barium-137. This seemed easy but to understand the concept of tiny beyond belief particles shooting from a substance until that substance becomes stable maybe in minutes or maybe in millions of years is amazing. This is the thing that I have come to appreciate about science, at a first glance the mol may seem like a tedious conversion process with a lot of zeros, but when you have a chance to stop and think about it you real in the wonder of how enormously eternal the pieces that make up the universe are. All in all processes can be interesting or they can be mind straining but the results are so important to humanity.
Through my scientific paper I learned that nuclear energy is very powerful. I learned that if we wish to use it at a greater scale in the future there are a lot of decisions that lay on our hands. One of these disiions for instance is that we need to look into the effects of elements with long half lives that will contaminate what is around them for a long time versus elements with short half lives that are very dangerous for a short amount of time. Through my infographic I learned that learned the amount of Ionizing radiation we have been exposed to has gone up by a considerable amount from medical procedures involving radiation. Here is another example of dilemma that nuclear power provides humanity with. Something that causes cancer can also help heal us from it.
Through my infographic I was trying to show people that we are exposed to a considerably higher amount of radiation then we were in 1987. I wanted to convey this so that people can understand not only what Ionizing radiation is, and what its effects are but also the reality of how we use it and what actions we are taking with it that both benefit and harm humanity. I wanted to provide an accurate representation of the Ionizing radiation sources we are exposed to. I wanted to show the benefits and the drawbacks of nuclear medicine and radiation therapy because those are the greatest growing categories of our exposure. I wanted to do this all so that people could have more understanding about our world and our interaction with it through our technologies.
Measuring Half-Life of Barium 137 after Processing Cesium-137 through an Isotopic Generator
Liam O’Brien, Nicholas Turco
ABSTRACT:
The purpose of this experiment was to come to a better understanding of the half-life curve of a radioactive substance specifically barium-137 and to observe the half-life curve in using the graphing software LOGGER PRO. The question of this experiment was to determine whether we could accurately observe the half-life curve of Barium-137 using an isogenerator, Geiger counter, and graphing software in order to understand what a half-life is and the importance of a half-life we viewed the exponential decay of a graph. Our method involved using an isogenorator to convert Cesium-137 into Barium-137 and to eject radioactive Barium-137 from the isogenorator to a measuring palate in which the decay of the Barium-137 could be measured. We recorded this decay and found that the overall curve of our results closely resembled the theoretical curve. We found that although the results of our experiment resembled half-life curves we had previously encountered; the decay in an experiment does not perfectly equate to the theoretical curve modeled by an equation. Through this experiment we learned that although our measurements can be very accurate, experimental data will never perfectly resemble theoretical data.
INTRODUCTION:
A radioactive substance is an element that emits ionizing radiation through radioactive decay. Ionizing radiation occurs when a particle has enough energy to displace an electron. Radioactive decay is the process in which an unstable nuclide (atomic nucleus) emits charged particles and/or electromagnetic radiation in order to conserve mass and energy and move towards greater overall entropy. Radioactive decay exists in the forms of alpha, beta and gamma decay. In an alpha decay an alpha particle is emitted; alpha particles are positively charged and identical to the nucleus of a Helium atom--with two protons and two neutrons. Beta decay is the second process of ionizing radiation. Beta decay takes place when there are too many neutrons in an atom for the amount of protons: thus a neutron splits into one electron and one proton. The third type of radioactive decay, gamma radiation, is the process of an over excited nucleus emitting a photon. Gamma decay frequently follows alpha and beta decay, as it does in our experiment. Cesium-137 decays to Barium-137, a beta decay, and is followed by the gamma decay of Barium-137.
All radioactive substances have different rates at which they decay; scientists distinguish this rate with the term half-life. A half-life is the average amount of time it takes for half of a sample of a specific isotope to decay. The half-lives of different elements differ drastically. For example: the half-life of Tellurium-128 is 7.7 x 1024 years and Barium-137 has a half-life of 2.552 minutes. The purpose of this Lab was to learn how to accurately measure the half-life of a sample of Barium-137.
MATERIALS:
Computer (self-provided)
Vernier computer interface (will be self-provided)
Logger Pro (will be self-provided)
Radiation Monitor
Cesium/Barium-137 Isotopic Generator
Small metal dish for Barium Solution
Eluting solution of HCl and NaCl
METHODS:
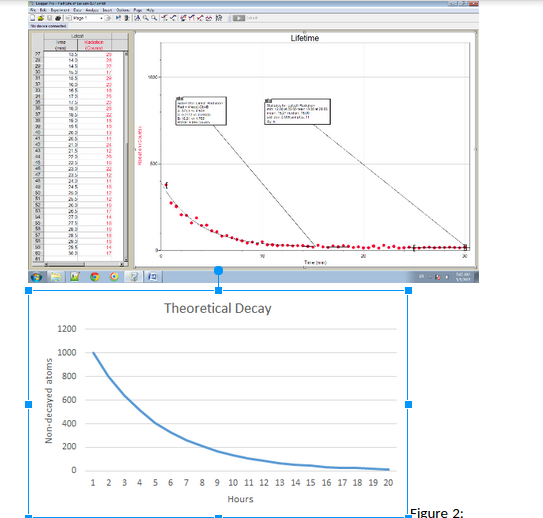
We first prepared a small metal dish with sides that were no more than 1 cm in height to receive the barium solution. We prepared the computer for data collection by opening the file “03 Lifetime” from the Nuclear Radiation w Vernier folder of Logger Pro. With this graph we counted rate vs. time and the exponential radioactive decay of Barium 137. The vertical axis was scaled from 0 to 1200 counts/interval. The horizontal axis, time, was scaled from 0 to 30 minutes. We then prepared the Isotopic Generator for use as directed by the manufacturer. We extracted the barium solution into the prepared dish. Then worked quickly from the time of solution extraction to the start of data collection, because the Barium-137 sample began to decay immediately. We placed the Geiger counter over the cup so that the counts it measured could be maximized. We then started the process of Logger Pro counting the number of gamma photons that struck the detector during each 30 second count interval. Data collection continued for 30 minutes. The detector and the barium cup remained motionless for the entirety of the process. After data collection was completed and all data was collected we set the radiation monitor aside, and disposed of the barium solution and cup following all safety precautions and procedures applying to our materials and chemical substances.
HAZARDS:
Cesium-137 is a radioactive nuclide that emits beta radiation. Working with a small amount of Cesium 137 for 2 hours means that a person will be exposing themselves to approximately .029 millirems of radiation. In a given year the average United States citizen experiences approximately 350 millirems of radiation. Barium- 137 is a radioactive nuclide with a half-life of 2.5 minutes, while decaying it emits one gamma ray. Barium 137 is a heavy alkaline earth metal and is toxic. Precautions and steps should be followed when working with the mentioned substances to ensure optimal safety. Wear Lab goggles for the entirety of the lab. Do not directly contact Cesium-137 sample. While working with these materials in the lab protection from radiation is not needed, long clothes and gloves may be worn. Emergency procedures for Cesium-137 are as follows. In case of personal contamination wash contaminated skin with water and soap and monitor skin for a short period afterward. Do not abrade skin instead blot dry. In the case of a spill or a leak alert everyone in the area, simultaneously clearing area. Summon aid and confine spill or leek to with absorbent materials. In case of damage to sealed radioactive source holder. First immediately evacuate the vicinity around the source holder. Next place a barrier at a safe distance from the source holder at least 5 meters. Finally Identify area as a radiation hazard and contact emergency number posted on local warning sign.
Suggested Emergency Protective Equipment include Gloves, Footwear. Covers Safety Glasses, as well as Outer layer or easily removed protective clothing (as situation requires).
RESULTS:
The count rate decreases over time. The decrease is consistent with the activity of counts proportional to the amount of radioactive material remaining. On average the counts decrease incrementally less as time goes on. In other words as the decay increases the activity and amount of counts decreases. This is due to the fact that over time, more Barium-137 decays, as this happens there are less radionuclides of barium left to decay.
Table 1:
equation for exponential decay
Y= A exp ( -C* X) +B
A
373.3 =+/- 8.509
B
16.21 = +/- 1.765
C
0.2772 = +/- 0.01003
λ (min^-1)
.2772
t-½ (min)
2.5
λ is the radioactive decay constant (the rate at which Barium-137 decays). The term t-½ is the half-life of Barium-137. The half-life is equal to .693/λ. The variable A is the initial amount of radiation when the decay stars.
The variable B accounts for the background radiation picked up by the Geiger Counter as well. The variable C is the decay constant and is equal to λ.
Liam O’Brien, Nicholas Turco
ABSTRACT:
The purpose of this experiment was to come to a better understanding of the half-life curve of a radioactive substance specifically barium-137 and to observe the half-life curve in using the graphing software LOGGER PRO. The question of this experiment was to determine whether we could accurately observe the half-life curve of Barium-137 using an isogenerator, Geiger counter, and graphing software in order to understand what a half-life is and the importance of a half-life we viewed the exponential decay of a graph. Our method involved using an isogenorator to convert Cesium-137 into Barium-137 and to eject radioactive Barium-137 from the isogenorator to a measuring palate in which the decay of the Barium-137 could be measured. We recorded this decay and found that the overall curve of our results closely resembled the theoretical curve. We found that although the results of our experiment resembled half-life curves we had previously encountered; the decay in an experiment does not perfectly equate to the theoretical curve modeled by an equation. Through this experiment we learned that although our measurements can be very accurate, experimental data will never perfectly resemble theoretical data.
INTRODUCTION:
A radioactive substance is an element that emits ionizing radiation through radioactive decay. Ionizing radiation occurs when a particle has enough energy to displace an electron. Radioactive decay is the process in which an unstable nuclide (atomic nucleus) emits charged particles and/or electromagnetic radiation in order to conserve mass and energy and move towards greater overall entropy. Radioactive decay exists in the forms of alpha, beta and gamma decay. In an alpha decay an alpha particle is emitted; alpha particles are positively charged and identical to the nucleus of a Helium atom--with two protons and two neutrons. Beta decay is the second process of ionizing radiation. Beta decay takes place when there are too many neutrons in an atom for the amount of protons: thus a neutron splits into one electron and one proton. The third type of radioactive decay, gamma radiation, is the process of an over excited nucleus emitting a photon. Gamma decay frequently follows alpha and beta decay, as it does in our experiment. Cesium-137 decays to Barium-137, a beta decay, and is followed by the gamma decay of Barium-137.
All radioactive substances have different rates at which they decay; scientists distinguish this rate with the term half-life. A half-life is the average amount of time it takes for half of a sample of a specific isotope to decay. The half-lives of different elements differ drastically. For example: the half-life of Tellurium-128 is 7.7 x 1024 years and Barium-137 has a half-life of 2.552 minutes. The purpose of this Lab was to learn how to accurately measure the half-life of a sample of Barium-137.
MATERIALS:
Computer (self-provided)
Vernier computer interface (will be self-provided)
Logger Pro (will be self-provided)
Radiation Monitor
Cesium/Barium-137 Isotopic Generator
Small metal dish for Barium Solution
Eluting solution of HCl and NaCl
METHODS:
We first prepared a small metal dish with sides that were no more than 1 cm in height to receive the barium solution. We prepared the computer for data collection by opening the file “03 Lifetime” from the Nuclear Radiation w Vernier folder of Logger Pro. With this graph we counted rate vs. time and the exponential radioactive decay of Barium 137. The vertical axis was scaled from 0 to 1200 counts/interval. The horizontal axis, time, was scaled from 0 to 30 minutes. We then prepared the Isotopic Generator for use as directed by the manufacturer. We extracted the barium solution into the prepared dish. Then worked quickly from the time of solution extraction to the start of data collection, because the Barium-137 sample began to decay immediately. We placed the Geiger counter over the cup so that the counts it measured could be maximized. We then started the process of Logger Pro counting the number of gamma photons that struck the detector during each 30 second count interval. Data collection continued for 30 minutes. The detector and the barium cup remained motionless for the entirety of the process. After data collection was completed and all data was collected we set the radiation monitor aside, and disposed of the barium solution and cup following all safety precautions and procedures applying to our materials and chemical substances.
HAZARDS:
Cesium-137 is a radioactive nuclide that emits beta radiation. Working with a small amount of Cesium 137 for 2 hours means that a person will be exposing themselves to approximately .029 millirems of radiation. In a given year the average United States citizen experiences approximately 350 millirems of radiation. Barium- 137 is a radioactive nuclide with a half-life of 2.5 minutes, while decaying it emits one gamma ray. Barium 137 is a heavy alkaline earth metal and is toxic. Precautions and steps should be followed when working with the mentioned substances to ensure optimal safety. Wear Lab goggles for the entirety of the lab. Do not directly contact Cesium-137 sample. While working with these materials in the lab protection from radiation is not needed, long clothes and gloves may be worn. Emergency procedures for Cesium-137 are as follows. In case of personal contamination wash contaminated skin with water and soap and monitor skin for a short period afterward. Do not abrade skin instead blot dry. In the case of a spill or a leak alert everyone in the area, simultaneously clearing area. Summon aid and confine spill or leek to with absorbent materials. In case of damage to sealed radioactive source holder. First immediately evacuate the vicinity around the source holder. Next place a barrier at a safe distance from the source holder at least 5 meters. Finally Identify area as a radiation hazard and contact emergency number posted on local warning sign.
Suggested Emergency Protective Equipment include Gloves, Footwear. Covers Safety Glasses, as well as Outer layer or easily removed protective clothing (as situation requires).
RESULTS:
The count rate decreases over time. The decrease is consistent with the activity of counts proportional to the amount of radioactive material remaining. On average the counts decrease incrementally less as time goes on. In other words as the decay increases the activity and amount of counts decreases. This is due to the fact that over time, more Barium-137 decays, as this happens there are less radionuclides of barium left to decay.
Table 1:
equation for exponential decay
Y= A exp ( -C* X) +B
A
373.3 =+/- 8.509
B
16.21 = +/- 1.765
C
0.2772 = +/- 0.01003
λ (min^-1)
.2772
t-½ (min)
2.5
λ is the radioactive decay constant (the rate at which Barium-137 decays). The term t-½ is the half-life of Barium-137. The half-life is equal to .693/λ. The variable A is the initial amount of radiation when the decay stars.
The variable B accounts for the background radiation picked up by the Geiger Counter as well. The variable C is the decay constant and is equal to λ.
Materials Unit project
The Significance of the Northeast Tanning Industry:
When gazed upon, much of the Northeast’s forests appear to be small hardwood leafed trees such as beech and cherry. When asked who had the superior equipment in the Civil War, most would reply the Union. When we look deeper into this iconic landscape and historic war, we will find among many factors, that the tanning industry of the late eighteenth and early nineteenth century played a role in shaping american history and landscape.
Through much of history tanning was considered an odorous practice and. It was largely performed at the edges of towns. primarily in poor communities during the middle ages. It consisted of removing the hair of off hides with urine while binding the the proteins in the actual tanning process with animal brains, feces, or fats.
However in the late eighteenth and early nineteenth centuries a change in the way that we tanned animal hides dramatically altered America’s people and its land. Vegetable tanning was a new method that was implemented at the time throughout the Northeast.
Vegetable tanning consists of using the tannin found in the bark, roots, and leaves of trees to bind with the Collagen Proteins that make up most of the connective tissue in animals, including ligaments fibrous tissue and skin. Collagens are long fibrils that make up 25%-35% of the whole body protein. Tannin is a kind of polyphenol astringent chemical compound that forms strong bonds with proteins and other macromolecules, it shrinks body tissue as well as makes it release water but not absorb it. Astringents are polymers that contain high amounts of hydroxyls and carboxyls. A carboxylic acid is a complex organic compound that contains a carboxyl group R-(c) O RH. This means that a carbon atom bonds two of its bonds with an oxygen atom. During the Tanning Process tannins bind to collagen proteins and shrink them, making them less water soluble as well as protect them from bacteria and give them flexibility.
Tannin is found in wine unripe fruit, tea and coffee giving your mouth a bitter puckered feeling; however the source of tannin that gave rise to the lucrative and explosive tanning industry of the nineteenth century was the hemlock tree. The bark of the hemlock holds a high percentage of tannin of 10-12%. The trees brak was harvested, cut, ground and then boiled, in order to extract its tannin. Tanneries were first established in the Catskills of New York because of the rich forests of hemlock and the expansive Hudson river which lay near by, beckoning as a railway for the shipping of hides.
Tanneries were a great source of revenue for their owners and provided harsh but steady work for many immigrants at the time. Many of the people that worked at tanneries also owned cattle and sheep that they sold to the industry. Tanneries would make towns, complete with churches, schools, stores, and houses spring up in their wake, in once wild territory. Tanneries also employed people as bark peelers. Bark peelers ussaly felled and then stripted hemlocks of their bark, leaving the rotting decay of pale trunks in the place of once mighty forests. The rate at which the tanning industry produced hides took its toll on the hemlock population and soon people were hard pressed to find find easily accessible tree supplies, leading to the life span of tanneries to be short and sporadic. The average amount of time that a tannery flourished was 30 years.
The civil war allowed this already successful industry to explode in production and wealth. The legacy of Sullivan County in the Catskills is a unique piece of history linked to Tanning. Almost all of the Unions leather gear such as harnesses, saddles, bags, and boots were tanned in Sullivan County. With 39 Tanneries Sullivan County had the highest number of tanneries than any other county at the time. This gear had a distinct red hew from the hemlock trees quality tannin.The Unions superior equipment in the Civil war was due to the North's Industrial Abilities. One of the industries that aided in providing the best equipment for the Union was the Tanning Industry. The speed at which tanning industries could produce leather greatly exceeded that of the confederacy. The quality of the hemlocks and their abundance was also instrumental in allowing the industry to supply soldiers with superior leather gear. Particular Quality equipment included hearty boots, and safe Carriage Boxes. Railroads such as the Erie railroad that were used to transport hides to tanneries also served as swift modes of transportation from union soldiers to and fro battles.
By the end of the hemlock tanning craze, 70 million trees had been harvested for their bark. A common misconception is that hemlocks only grow in swamps or high mountainous regions, the truth is that hemlocks that grow in these places were not as accessible to bark peelers and so remain more intact in these Landscapes.The trunks left behind created major forest fires. Now much of the landscapes that used to be a rolling expanse of green hemlocks are either populated with short leafed hardwood trees such as beech and cherry or still remain barren and slowly recovering. There is only one virgin hemlock tree standing in the Catskills today.
In the industry’s heyday there were at least thirty major forest fires in thirty years, amounting to a major forest fire each year. Another environmental hazard that the tanning industry created during the rise of the industrial use of vegetable tanning was the pollutants such as tanning liquors, limes and animal parts were dumped into rivers. in the end the massive rise of the tanning industry had an equally rapid demise as its rise.
By the end of the nineteenth century the tanning industry was gone with the mighty hemlock. After chrome tanning, a method that used minerals and synthetic chemicals to create the tanning process as well as alternative substances such as nylon and polypropylene. However the process of vegetable tanning forever shaped American history and landscape.
Reflection on material unit project:
I have learned through this project that the chemistry of materials have shaped the past and maybe can shape the future in complex and deep ways. I decided to study the tanning industry of the Northeast in the nineteenth century. I learned that when people were able to use the tannin in hemlock trees to alter raw hide into leather at a mass scale the land of the united states and its history was changed. The ability that tannin in trees gave the north in mass producing leather gave unions much better equipment in the civil war and contributed to their success. However harvesting of hemlocks for their tannin has also shaped the Northeasts forest in a much different way then today. Finally i have learned that chemical changes in the way that we tan leather has the ability to change the future. if we can develop a method to tan leather that does not discharge toxic waste into the environment such as chrome tanning does, we could change out future for the better.
Vegetable tanning consists of using the tannin found in the bark, roots, and leaves of trees to bind with collagen Proteins that make up most of the connective tissue in animals, including ligaments fibrous tissue and skin. Collagen are made of long fibrils and make up 25%-35% of the whole body protein. Tannin is a kind of poly phenol astringent chemical. Astringent is a chemical compound that forms strong bonds with proteins and other macro molecules, it shrinks body tissue as well as makes it release water but not absorb it. Astringents are polymers that contain high amounts of hydroxyls and carboxyls. A carboxylic acid is a complex organic compound that contains a carboxyl group. R -(C) O RH. This means that a carbon atom bonds two of its bonds with an oxygen atom, while one more is bonded to another oxygen atom whose other bond is to a hydrogen atom. During the tanning process tannin binds to collagen proteins and shrink them, making them less water soluble as well as protect them from bacteria and give them flexibility. This is the way that tannin in hemlock trees and the relationship with the structure of collagen proteins in animal hides are determined on the atomic molecular, microscopic and macroscopic level.
Vegetable tanning consists of using the tannin found in the bark, roots, and leaves of trees to bind with collagen Proteins that make up most of the connective tissue in animals, including ligaments fibrous tissue and skin. Collagen are made of long fibrils and make up 25%-35% of the whole body protein. Tannin is a kind of poly phenol astringent chemical. Astringent is a chemical compound that forms strong bonds with proteins and other macro molecules, it shrinks body tissue as well as makes it release water but not absorb it. Astringents are polymers that contain high amounts of hydroxyls and carboxyls. A carboxylic acid is a complex organic compound that contains a carboxyl group. R -(C) O RH. This means that a carbon atom bonds two of its bonds with an oxygen atom, while one more is bonded to another oxygen atom whose other bond is to a hydrogen atom. During the tanning process tannin binds to collagen proteins and shrink them, making them less water soluble as well as protect them from bacteria and give them flexibility. This is the way that tannin in hemlock trees and the relationship with the structure of collagen proteins in animal hides are determined on the atomic molecular, microscopic and macroscopic level.
Elevator Pitch
Have you ever asked yourself why things are the way they are? Our lives are permeated with detail, history, and meaning that escape the common routine of our daily lives. Much of our environment, struggles, and benefits are connected to human advancement and ingenuity that in a large quantity remain overlooked and under-scrutinized by many the ordinary observer. As further illustration, take the Tanning industry of the early eighteenth and late nineteenth century. Would you have guessed that the simple shift from medieval odorous practices to using the tannins in the bark of hemlock trees to tan leather would forever shape Northeastern forests and the success of the Union in the Civil War? In my latest Book Pull Yourself Up By Your Bootstraps: A compendium of obscure and seldom- heeded material advancements in American society, you will find yourself devouring pages to understand just how and why you are here in the way you are. To enlighten yourself through this fundamental and instrumental history, invest in Nicholas Turco and Pull Yourself Up By Your Bootstraps, on your way out!
Have you ever asked yourself why things are the way they are? Our lives are permeated with detail, history, and meaning that escape the common routine of our daily lives. Much of our environment, struggles, and benefits are connected to human advancement and ingenuity that in a large quantity remain overlooked and under-scrutinized by many the ordinary observer. As further illustration, take the Tanning industry of the early eighteenth and late nineteenth century. Would you have guessed that the simple shift from medieval odorous practices to using the tannins in the bark of hemlock trees to tan leather would forever shape Northeastern forests and the success of the Union in the Civil War? In my latest Book Pull Yourself Up By Your Bootstraps: A compendium of obscure and seldom- heeded material advancements in American society, you will find yourself devouring pages to understand just how and why you are here in the way you are. To enlighten yourself through this fundamental and instrumental history, invest in Nicholas Turco and Pull Yourself Up By Your Bootstraps, on your way out!